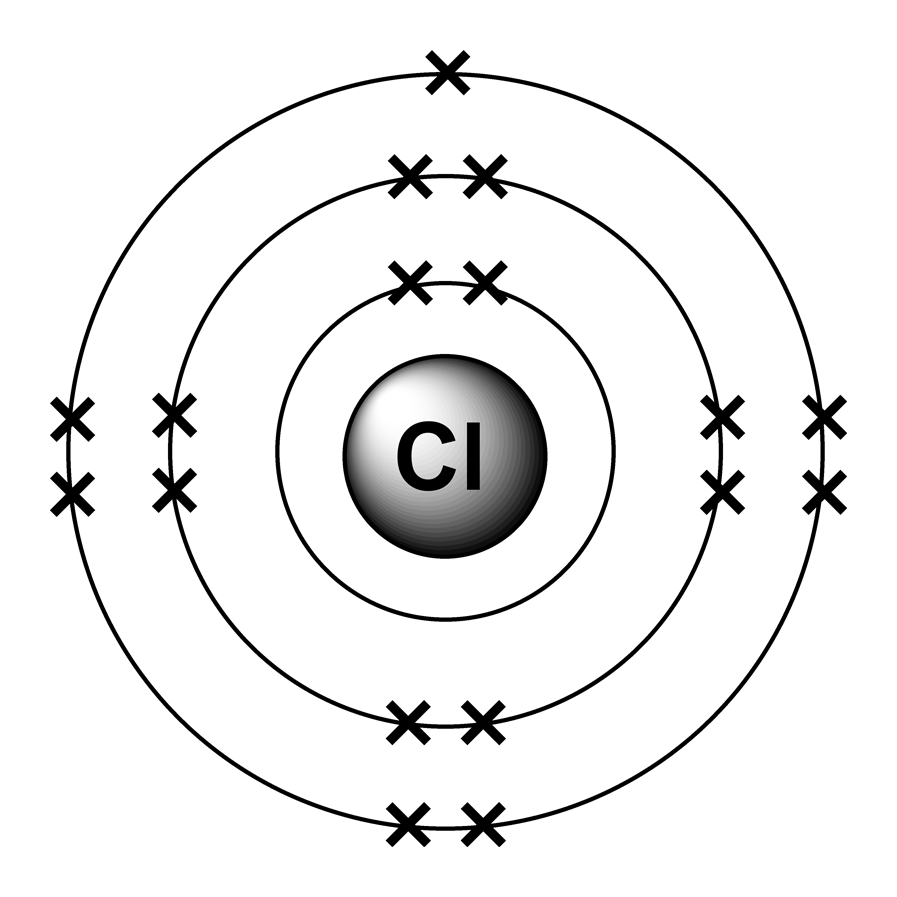
Neutrons can be added or removed from an atom and the electrons around the atom really don't care that much. Neutrons don't change things greatly because they do not carry an electrical charge. Adding or removing protons changes the number and arrangement of electrons in the outer shell which changes how that atom reacts with other atoms. The number of electrons in an atom's outer shell determines the atom's chemical properties. Each proton in an atom's nucleus must be balanced with a negatively charged electron in one of the 'shells' outside the nucleus. Why does changing the number of protons in an atom change which element that atom is but changing the number of neutrons doesn't? Protons carry a positive charge. An atom of chlorine-35 contains 18 neutrons (17 protons + 18 neutrons = 35 particles in the nucleus) while an atom of chlorine-37 contains 20 neutrons (17 protons + 20 neutrons = 37 particles in the nucleus).Īdding or removing a neutron from an atom's nucleus creates isotopes of a particular element. Since all atoms of chlorine contain 17 protons, chlorine-35 and chlorine-37 differ in the number of neutrons each one has. Atomic mass indicated on entries of the Periodic Table. On the other hand, the atomic number (Z) of each element is found above the atomic symbol. The atomic mass of tin (Sn) is 118.71 u while the atomic mass of carbon (C) is 12.011 u. A property closely related to an atom’s mass number is its atomic mass. If you want to calculate how many neutrons an atom has, you can simply subtract the number of protons, or atomic number, from the mass number. 
The mass number is a tally of the number of protons and the number of neutrons in an atom's nucleus. The atomic mass of each element is found under the element symbol in the periodic table. Together, the number of protons and the number of neutrons determine an element’s mass number: mass number protons + neutrons. The number after the name 'chlorine' is called the mass number. Adding or removing a proton from an atom's nucleus changes that atom's atomic number and creates a different element.Ĭhlorine-35 and chlorine-37 are both isotopes of the element chlorine. Chlorine is in the halogen group (17) and is the second lightest halogen following fluorine. All atoms which contain 17 protons are called chlorine atoms. It also has a relative atomic mass of 35.5. The number of protons an atom has, also known as the atom's atomic number, determines which element it is. Atomic Number Atomic Mass Electron Configuration Number of Neutrons Melting Point Boiling Point Date of Discovery Crystal Structure. The isotope of carbon that has 6 neutrons is therefore C612.Why aren't Chlorine-35 and Chlorine-37 two different elements?Ĭhlorine-35 and chlorine-37 are not different elements because an atom of chlorine-35 and an atom of chlorine-37 each contain the same number of protons. An isotope of any element can be uniquely represented as XZA, where X is the atomic symbol of the element. In a typical sample of carbon-containing material, 98.89% of the carbon atoms also contain 6 neutrons, so each has a mass number of 12. The element carbon (C) has an atomic number of 6, which means that all neutral carbon atoms contain 6 protons and 6 electrons. Two moles of chlorine atoms are formed by breaking the bonds in a mole of chlorine. The isotopes of an element differ only in their atomic mass, which is given by the mass number ( A), the sum of the numbers of protons and neutrons. chlorine by dividing the mass of copper by the mass of chlorine. Atomic mass of Chlorine (Cl) 35.45: 35: 18: Atomic mass of Argon. All isotopes of an element have the same number of protons and electrons, which means they exhibit the same chemistry. Atomic mass of all elements (along with the rounded off values) is mentioned in the chart below. Atoms that have the same number of protons, and hence the same atomic number, but different numbers of neutrons are called isotopes. However, if you take any chlorine atom, it will not have this molar mass because this molar is the. 
Unlike protons, the number of neutrons is not absolutely fixed for most elements. By calculating, a chlorine atom has molar mass of 35.5g/mole. Recall from Section 1.6 that the nuclei of most atoms contain neutrons as well as protons. German for “wolf stone” because it interfered with the smelting of tin and was thought to devour the tin \) Element Symbols Based on Names No Longer in Use Elementįrom Cyprium, Latin name for the island of Cyprus, the major source of copper ore in the Roman Empire
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
